Environmental, Social and Governance (ESG)

Environmental, Social and Governance (ESG)
ESG
As a global leader in joint preservation, we make it our mission to deliver innovative, high-quality orthopedic solutions that restore active living for people around the world. Our clinician customers and their patients are at the heart of everything we do. But we also believe that we have a broader responsibility to take into account the positive effect we seek to have on our employees, and the environment and the communities where we live and work.
Our ESG Focus
In 2021, with the support and guidance from our Board of Directors, we embarked on a process to develop a foundational Environmental, Social and Governance (ESG) framework for our organization. This framework integrates our six key corporate values: People, Quality, Integrity, Accountability, Innovation and Teamwork. The initial step in our ESG journey included the completion of a “materiality assessment” based on the Sustainability Accounting Standards Board (SASB) framework. Our materiality assessment was a research-intensive and stakeholder-inclusive process and included guidance and insight from external advisors, and crucial feedback from key internal and external stakeholders, including investors, customers, suppliers, employees and members of our Board.
As a result of the materiality assessment, we identified the themes that are most important to our stakeholders and our business within traditional environmental, social and governance pillars. Most immediately, our materiality assessment enabled us to select our six key focus areas, with a goal to be aligned with SASB standards for the Medical Device industry. Each of our ESG focus areas will have senior management team sponsorship and engagement. We will continue to assess and update our ESG initiatives as our business grows and as we implement processes and improvements over time. The materiality assessment, and the initiatives that have resulted from it, have been undertaken with full support and oversight of the Board.

Environmental
Social
Governance

EcoVadis Sustainability Recognition
In 2022, our progress in ESG earned us the Silver Medal in Sustainability from EcoVadis, a leading provider of sustainability assessments for businesses worldwide. The Silver Medal from EcoVadis signifies that Anika scored in the top 25th percentile of companies assessed in 2022.
For more information about our EcoVadis rating and scorecard, please contact Mark Namaroff, VP of Investor Relations, ESG and Corporate Communications, at mnamaroff@anika.com.
Our Mission and Values
Powered by our passionate team, we partner with clinicians to create and provide meaningful advancements in early-intervention orthopedic care. We are unwavering in our commitment to quality and compliance as we develop and commercialize solutions that restore active living for people around the world.

Our ESG Focus Areas
Our Environmental Focus
As a global company, we recognize the importance of reducing the impact of our operations on the environment. Though Anika is not a significant generator of greenhouse gases (GHG), we are continually looking for ways to reduce energy consumption and to minimize GHG in our atmosphere.
Protecting the Environment: Climate, Energy Use and Emissions
Environmental Performance and Goals
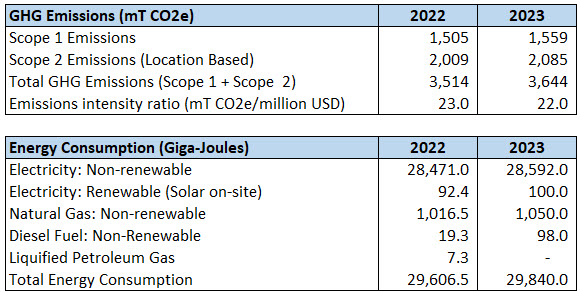 In 2023, Anika committed to evaluate ways to reduce its carbon footprint in its contribution to the Paris Climate agreement which seeks to limit global warming to 1.5o C and to achieve carbon neutrality by 2050. The first step in our process was to establish a baseline for GHG emissions by measuring Scope 1 and Scope 2 emissions across our global operations. Anika is a low GHG emitter due to our relatively small operational footprint. That said, we are committed to evaluating carbon reduction opportunities over the coming years in-line with our business priorities.
In 2023, Anika committed to evaluate ways to reduce its carbon footprint in its contribution to the Paris Climate agreement which seeks to limit global warming to 1.5o C and to achieve carbon neutrality by 2050. The first step in our process was to establish a baseline for GHG emissions by measuring Scope 1 and Scope 2 emissions across our global operations. Anika is a low GHG emitter due to our relatively small operational footprint. That said, we are committed to evaluating carbon reduction opportunities over the coming years in-line with our business priorities.
Reducing Energy Usage
In recent years, we have taken proactive steps to increase energy efficiency at our facilities. In 2020, for example, we completed the full transition to LED lighting at our Bedford, Massachusetts facility. Lighting upgrades were performed in four scopes, totaling approximately 373,500 kilowatts saved and an annualized cost savings of approximately $33,500 over the prior year.
We also installed automatic closing sashes to improve the ventilation of our workstation fume hoods, which are ventilated enclosures used to contain and exhaust gases. In addition to lowering electricity usage, the upgrade reduced natural gas consumption and emissions.
Implementing Proactive Recycling

In 2018, we began recycling the packaging materials used in the manufacture of our products, as well as paper goods used across the company. As a result of this program, in 2021 we diverted more than 6,000 pounds of recyclable materials from landfills, saving the equivalent of 54 trees, nine cubic yards of landfill, six barrels of oil and 21,000 gallons of water. We also continually look for ways to reduce waste across our business.
Conserving and Protecting Water
Water is a precious resource which we conserve and protect in several ways. We conduct rigorous testing and analysis at our Bedford, Massachusetts facility to prevent groundwater pollution and protect aquatic life. Our Bedford facility, which is adjacent to wetlands, uses state-of-the-art technology to trap sediment and other materials. We maintain storm water pollution plans and ensure that harmful chemicals, such as certain pesticides, are not used on the grounds of the facility. We also employ deionization technology to remove impurities in wastewater, thereby allowing the water to be reused during our manufacturing processes.
Our Social Focus
Human Capital: Diversity and Inclusion

We are committed to being a diverse, equitable and inclusive workplace where all employees, regardless of gender, race, ethnicity, national origin, age, sexual orientation or identity, education or disability are valued, respected and supported.
Promoting diversity throughout our company, including within the Board of Directors and our management team, has been a focal point of our Board of Directors, starting with the appointment of Cheryl R. Blanchard, Ph.D. as our President and Chief Executive Officer in early 2020. Currently, women account for approximately 40% our senior management team and our Board of Directors. Our seven-member Board currently includes three women and one person of color. View our 2022 proxy statement here for a description of our Board of Directors diversity matrix.
~40% of Anika's management team & Board of Directors are women
In 2020, Dr. Blanchard, with the support of our Board of Directors, joined other CEOs in signing the Massachusetts Biotechnology Council (MassBio) “CEO Pledge for a More Equitable and Inclusive Life Sciences Industry.” The signers of this pledge, over 220 CEOs, “recognize that racial inequity exists in [the life sciences] industry and in [life sciences] companies, and [they] must take responsibility to fix that injustice through comprehensive equity, diversity, and inclusion initiatives that are broad in scope, specific in action, and measurable in results.” The full text of the pledge can be found at: https://www.massbio.org/initiatives/equity-diversity-and-inclusion/open-letter/. As part of our commitment to the pledge, in 2021 we implemented a corporate Diversity, Equity and Inclusion (DEI) Policy Statement for inclusion in our employee handbook and new employee on-boarding process as well as created an internal Diversity Dashboard. The dashboard, which tracks diversity trends within every level of the organization, is shared with the Board of Directors quarterly, ensuring oversight at the highest level. The Anika DEI policy statement clearly conveys our commitment:
Anika is committed to an equitable, diverse, and inclusive workplace where all employees, regardless of their gender, race, ethnicity, national origin, age, sexual orientation or identity, education or disability are valued, respected, and supported. We will not discriminate based on these characteristics and will provide equal opportunities for employment and advancement throughout our company. Anika respects and appreciates diverse life experiences and heritages and will always work to ensure that all voices are heard and valued.
To complement these efforts, we are focused on broadening the level of diversity across the organization. Management compensation in 2022 is linked to specific DEI objectives, including continued progress on Anika’s MassBio Pledge and Anika's DEI initiatives such as implementation of a DEI employee survey, implementation of educational programs across the business including, but not limited to, topics such as unconscious bias and tone, and the importance of diversity at our company, establishing employee resource group(s), conducting a diversity audit on hiring processes, and implementing metrics to drive improvement around our DEI initiatives, processes and objectives.
Recognizing the value of employee retention and engagement, we conduct regular, company-wide employee engagement surveys. Our management team evaluates the results and identifies opportunities for improvement. As a result of this feedback, we established an employee led Communications Task Force chartered with improving communications across the business, and we launched a new e-learning program to expand employee development.
Social Capital: Product Quality and Safety

As a company that provides medical products to clinicians to treat their patients, product quality is of the utmost importance to ensure that Anika’s products are safe and effective to use in compliance with global regulatory requirements. Toward that end, we maintain a robust Quality Management System (QMS) that fosters continuous improvement, drives manufacturing excellence and ensures compliance with global standards. Comprehensive training for all employees enables us to consistently produce safe, high-quality products that meet or exceed the expectations of our customers. Quality is a core value of all employees at Anika.
As a global medical device manufacturer, we adhere to ISO 13485, an international standards for medical device Quality Management Systems with oversight provided by Notified Bodies GMED and BSi. Our QMS is also aligned with regulations from the U.S. Food and Drug Administration for medical device quality management systems.
Human Capital: Safe and Healthy Workplace
We provide resources, programs and services to support the physical, emotional and financial health of our employees. Additionally, specifically in regard to workplace safety, we have a Health, Safety and Environmental (HSE) Policy that all employees are required to be trained on, which can be viewed here.
As part of our robust workplace safety training, we have implemented interactive virtual learning systems. These systems are function-specific and ensure that all employees have access to critical information to keep them and their co-workers safe. We also use an electronic management system (EMS) implemented in 2018 to obtain information on any accidents or other safety issues. Our EMS offers the ability for anyone at the company to submit information about potential safety risks. All employees participate in annual training on workplace safety and the use of systems, including the EMS, to ensure that the importance of workplace safety is top of mind for employees throughout our company.
Our Governance Focus
A workplace culture of “do the right thing,” guided by high standards and comprehensive policies, is the bedrock to our success as a life science company. This principle underpins our ability to deliver life-changing solutions to our customers ─ and the patients they treat ─ around the world.
At Anika, a commitment to ethics and quality, as realized and demonstrated through personal integrity and accountability, forms the basis for everything
we do and forges the path to all that we can achieve. These foundational tenets of our company are non-negotiable and do not take a day off.
—Cheryl R. Blanchard, PhD
Governance: Business Ethics and Integrity

Our Code of Business Conduct and Ethics guides employees and the Board of Directors in making ethical and legal decisions when conducting business and performing their day-to-day duties. We also have Conflict of Interest and Insider Trading Policies, as well as a channel for employees and directors to report concerns through our Whistle Blower Policy. Please view our Corporate Governance documents webpage here for a full list of our governance documents and policies.
Our Compliance Program is based on our Code of Business Conduct and Ethics and is structured to help our company comply with the myriad of laws, guidelines and regulations that impact our business by mirroring the recommendations of the U.S. Department of Health and Human Services, Office of Inspector General’s (OIG’s) seven elements of an effective compliance program. We have established a Compliance Committee, made up of our Chief Executive Officer and senior representatives from several other functions within our company, which oversees the operation of our healthcare Compliance Program. In addition, we have established an Engagements Committee, which reviews and approves proposed interactions with healthcare professionals and healthcare organizations, including consulting services, royalty arrangements, requests for grants and research engagements. We are also a member of the Advanced Medical Technology Association, or AdvaMed, a global trade association of companies that develop, produce, manufacture, and market medical technologies, and which are dedicated to advancing medical science, developing high quality, innovative medical technology, and improving patient care. We follow AdvaMed’s Code of Ethics on Interactions with U.S. Health Care Professionals, or the AdvaMed Code of Ethics, which provides medical technology companies with guidance on ethical interactions and relationships with health care professionals. To view the AdvaMed Code of Ethics, please visit https://www.advamed.org/member-center/resource-library/advamed-code-of-ethics/.
In 2021, we also expanded our focus on working to ensure that our international distributors are organizations that operate in compliance with our policies and procedures and all laws and regulations. Using an independent screening process and third-party service providers that focus on investigating the integrity, reputation, and business practices of our potential distribution partners, we attempt to gauge the level of risk posed by international distributors based on their geographic location and past corporate behavior, among other factors, and tailor our due diligence efforts and our willingness to engage specific distributors accordingly. We recognize that vigilant oversight of this aspect of our supply chain is foundational to our ability to grow our business with high ethical standards and integrity.
Governance: Cybersecurity
In today’s business landscape, a robust cybersecurity program is crucial. We rely on a layered approach (defense-in-depth) to mitigate the constantly evolving cyber threats and continually invest in people, processes, and next-generation cybersecurity technologies to support this. Our global approach aligns with industry best practices and is grounded in the NIST framework, ensuring that the confidentiality, integrity, and availability of our systems, data, and products are protected. We follow a programmatic approach that involves leveraging trusted technology partners and solutions, independent assessments and testing, and user education, with risks tracked and key controls mapped to these risks to ensure that our security program adapts to the changing threat landscape. We also ensure that all third- party vendor management, governance, and data protection programs align with best practices, regulations, and standards to reduce organizational risk.
To manage cybersecurity incidents, our global security operations team executes our cybersecurity incident response plan, conducts regular readiness exercises, and continuously improves the program to manage the changing threats faced in our sector. We have also implemented an ongoing mandatory cybersecurity training program for all employees with routine attack simulations that has raised awareness of cyber threats for our users and our team members and are regularly updated on best practices to help reduce risks. The cybersecurity program is led by Anika’s Vice President of Information Technology with senior management team sponsorship and Audit Committee oversight.
Business Model: Supply Chain Management
Supply chain management is a fundamental aspect of our business to ensure we have the raw materials and equipment/parts available to manufacture our products in a timely fashion for our customers and their patients. We are successfully taking steps to navigate the challenges of the global supply chain crisis, including finding additional/alternative suppliers and reengineering our manufacturing processes and supplies for a more streamlined procurement process. Many of these supply chain improvements and the processes we’ve initiated to develop and implement them, though born of near-term necessity, are permanent and will enhance our ability to procure supplies to satisfy the needs of our customers and their patients as we continue to grow and transform our business. We recognize that vigilant oversight of the supply chain is important and are developing a new “Supplier Code of Conduct” that will further strengthen our oversight in this area.
For more information about Anika’s ESG Initiatives please contact
Matt Hall
Director, Corporate Development and Investor Relations
investorrelations@anika.com
